- What is an atom? Nucleus, protons and electrons
- Atomic particles
- Nucleus
- Protons
- Electrons
- Neutrons
- History of the atom
- Atoms
- Contents
- What is an atom?
- What are the parts of an atom?
- What is the Periodic Table?
- How do atoms make molecules and compounds?
- What are isotopes?
- How do atoms make ions?
- How many atoms are there in something?
- How do we know atoms exist?
- A brief history of atoms
- Find out more
- On this website
- On other sites
- Articles
- Books for younger readers
- Books for older readers
- Videos
- Follow us
- Rate this page
- Save or share this page
What is an atom? Nucleus, protons and electrons
By Tim Sharp 11 September 2019
Atoms are made up of a nucleus, protons and electrons.
Atoms are the basic units of matter and the defining structure of elements. The term «atom» comes from the Greek word for indivisible, because it was once thought that atoms were the smallest things in the universe and could not be divided. We now know that atoms are made up of three particles: protons, neutrons and electrons — which are composed of even smaller particles, such as quarks.
Atoms were created after the Big Bang 13.7 billion years ago. As the hot, dense new universe cooled, conditions became suitable for quarks and electrons to form. Quarks came together to form protons and neutrons, and these particles combined into nuclei. This all took place within the first few minutes of the universe’s existence, according to CERN.
It took 380,000 years for the universe to cool enough to slow down the electrons so that the nuclei could capture them to form the first atoms. The earliest atoms were primarily hydrogen and helium, which are still the most abundant elements in the universe, according to Jefferson Lab. Gravity eventually caused clouds of gas to coalesce and form stars, and heavier atoms were (and still are) created within the stars and sent throughout the universe when the star exploded (supernova).
Atomic particles
Protons and neutrons are heavier than electrons and reside in the nucleus at the center of the atom. Electrons are extremely lightweight and exist in a cloud orbiting the nucleus. The electron cloud has a radius 10,000 times greater than the nucleus, according to the Los Alamos National Laboratory.
Protons and neutrons have approximately the same mass. However, one proton is about 1,835 times more massive than an electron. Atoms always have an equal number of protons and electrons, and the number of protons and neutrons is usually the same as well. Adding a proton to an atom makes a new element, while adding a neutron makes an isotope, or heavier version, of that atom.
Nucleus
The nucleus was discovered in 1911 by Ernest Rutherford, a physicist from New Zealand. In 1920, Rutherford proposed the name proton for the positively charged particles of the atom. He also theorized that there was a neutral particle within the nucleus, which James Chadwick, a British physicist and student of Rutherford’s, was able to confirm in 1932.
Virtually all the mass of an atom resides in its nucleus, according to Chemistry LibreTexts. The protons and neutrons that make up the nucleus are approximately the same mass (the proton is slightly less) and have the same angular momentum, or spin.
The nucleus is held together by the strong force, one of the four basic forces in nature. This force between the protons and neutrons overcomes the repulsive electrical force that would otherwise push the protons apart, according to the rules of electricity. Some atomic nuclei are unstable because the binding force varies for different atoms based on the size of the nucleus. These atoms will then decay into other elements, such as carbon-14 decaying into nitrogen-14.
Protons
Protons are positively charged particles found within atomic nuclei. Rutherford discovered them in experiments with cathode-ray tubes that were conducted between 1911 and 1919. Protons are about 99.86% as massive as neutrons.
The number of protons in an atom is unique to each element. For example, carbon atoms have six protons, hydrogen atoms have one and oxygen atoms have eight. The number of protons in an atom is referred to as the atomic number of that element. The number of protons also determines the chemical behavior of the element. Elements are arranged in the Periodic Table of the Elements in order of increasing atomic number.
Three quarks make up each proton — two «up» quarks (each with a two-thirds positive charge) and one «down» quark (with a one-third negative charge) — and they are held together by other subatomic particles called gluons, which are massless.
Electrons
Electrons are tiny compared to protons and neutrons, over 1,800 times smaller than either a proton or a neutron. Electrons are about 0.054% as massive as neutrons, according to Jefferson Lab.
Joseph John (J.J.) Thomson, a British physicist, discovered the electron in 1897, according to the Science History Institute. Originally known as «corpuscles,» electrons have a negative charge and are electrically attracted to the positively charged protons. Electrons surround the atomic nucleus in pathways called orbitals, an idea that was put forth by Erwin Schrödinger, an Austrian physicist, in the 1920s. Today, this model is known as the quantum model or the electron cloud model. The inner orbitals surrounding the atom are spherical but the outer orbitals are much more complicated.
An atom’s electron configuration refers to the locations of the electrons in a typical atom. Using the electron configuration and principles of physics, chemists can predict an atom’s properties, such as stability, boiling point and conductivity, according to the Los Alamos National Laboratory.
Neutrons
The neutron’s existence was theorized by Rutherford in 1920 and discovered by Chadwick in 1932, according to the American Physical Society. Neutrons were found during experiments when atoms were shot at a thin sheet of beryllium. Subatomic particles with no charge were released – the neutron.
Neutrons are uncharged particles found within all atomic nuclei (except for hydrogen). A neutron’s mass is slightly larger than that of a proton. Like protons, neutrons are also made of quarks — one «up» quark (with a positive 2/3 charge) and two «down» quarks (each with a negative one-third charge).
History of the atom
The theory of the atom dates at least as far back as 440 B.C. to Democritus, a Greek scientist and philosopher. Democritus most likely built his theory of atoms upon the work of past philosophers, according to Andrew G. Van Melsen, author of «From Atomos to Atom: The History of the Concept Atom» (Duquesne University Press, 1952).
Democritus’ explanation of the atom begins with a stone. A stone cut in half gives two halves of the same stone. If the stone were to be continuously cut, at some point there would exist a piece of the stone small enough that it could no longer be cut. The term «atom» comes from the Greek word for indivisible, which Democritus concluded must be the point at which a being (any form of matter) cannot be divided any more.
His explanation included the ideas that atoms exist separately from each other, that there are an infinite amount of atoms, that atoms are able to move, that they can combine together to create matter but do not merge to become a new atom, and that they cannot be divided, according to Universe Today. However, because most philosophers at the time — especially the very influential Aristotle — believed that all matter was created from earth, air, fire and water, Democritus’ atomic theory was put aside.
John Dalton, a British chemist, built upon Democritus’ ideas in 1803 when he put forth his own atomic theory, according to the chemistry department at Purdue University. Dalton’s theory included several ideas from Democritus, such as atoms are indivisible and indestructible and that different atoms form together to create all matter. Dalton’s additions to the theory included the following ideas: That all atoms of a certain element were identical, that atoms of one element will have different weights and properties than atoms of another element, that atoms cannot be created or destroyed and that matter is formed by atoms combining in simple whole numbers.
Thomson, the British physicist who discovered the electron in 1897, proved that atoms can be divided, according to the Chemical Heritage Foundation. He was able to determine the existence of electrons by studying the properties of electric discharge in cathode-ray tubes. According to Thomson’s 1897 paper, the rays were deflected within the tube, which proved that there was something that was negatively charged within the vacuum tube. In 1899, Thomson published a description of his version of the atom, commonly known as the «plum pudding model.» An excerpt of this paper is found on the Chem Team site. Thomson’s model of the atom included a large number of electrons suspended in something that produced a positive charge giving the atom an overall neutral charge. His model resembled plum pudding, a popular British dessert that had raisins suspended in a round cake-like ball.
The next scientist to further modify and advance the atomic model was Rutherford, who studied under Thomson, according to the chemistry department at Purdue University. In 1911, Rutherford published his version of the atom, which included a positively charged nucleus orbited by electrons. This model arose when Rutherford and his assistants fired alpha particles at thin sheets of gold. An alpha particle is made up of two protons and two neutrons, all held together by the same strong nuclear force that binds the nucleus, according to the Jefferson Lab.
The scientists noticed that a small percentage of the alpha particles were scattered at very large angles to the original direction of motion while the majority passed right through hardly disturbed. Rutherford was able to approximate the size of the nucleus of the gold atom, finding it to be at least 10,000 times smaller than the size of the entire atom with much of the atom being empty space. Rutherford’s model of the atom is still the basic model that is used today.
Several other scientists furthered the atomic model, including Niels Bohr (built upon Rutherford’s model to include properties of electrons based on the hydrogen spectrum), Erwin Schrödinger (developed the quantum model of the atom), Werner Heisenberg (stated that one cannot know both the position and velocity of an electron simultaneously), and Murray Gell-Mann and George Zweig (independently developed the theory that protons and neutrons were composed of quarks).
Additional resources:
- Read more about the early universe, from CERN.
- Learn more about the history of atomic chemistry in this video from Khan Academy.
- Check out this simple slide show about atoms from the Jefferson Lab.
This article was updated on Sept. 10, 2019, by Live Science contributor Traci Pedersen.
Источник
Atoms
by Chris Woodford . Last updated: October 8, 2021.
S uppose you had to build yourself a world exactly like the one we live in. Where would you start? You’d need people. cars. houses. animals. trees. and billions of other things. But if you had a few dozen different types of atom , you could build all these things and more: you’d just join the atoms together in different ways. Atoms are the tiny building blocks from which everything around us is constructed. It’s amazing to think you can make anything out of atoms, from a snake to an ocean liner—but it’s absolutely true! Let’s take a closer look.
Artwork: From the hair on your head to the t-shirt on your back, everything in the world is made of atoms. I’ve greatly exaggerated their size in this illustration. On my screen, each of the atomic red dots is about 10 million times bigger than a typical atom. (Your screen may be bigger or smaller than mine, or scaled differently, so take that as a very rough approximation.)
Contents
What is an atom?
Take anything apart and you’ll find something smaller inside. There are engines inside cars, pips inside apples, hearts and lungs inside people, and stuffing inside teddy bears. But what happens if you keep going? If you keep taking things apart, you’ll eventually, find that all matter (all the «stuff» that surrounds us) is made from different types of atoms. Living things, for example, are mostly made from the atoms carbon, hydrogen, and oxygen. These are just three of over 100 chemical elements that scientists have discovered. Other elements include metals such as copper, tin, iron and gold, and gases like hydrogen and helium. You can make virtually anything you can think of by joining atoms of different elements together like tiny LEGO® blocks.
Photo: What does an atom look like? You can see one if you have the right kind of microscope or camera! This photo shows strontium atoms «flying» in a cube while being stimulated with precision laser light. By courtesy of National Institute of Standards and Technology (NIST).
An atom is the smallest possible amount of a chemical element—so an atom of gold is the smallest amount of gold you can possibly have. By small, I really do mean absolutely, nanoscopically tiny: a single atom is hundreds of thousands of times thinner than a human hair, so you have absolutely no chance of ever seeing one unless you have an incredibly powerful electron microscope. In ancient times, people thought atoms were the smallest possible things in the world. In fact, the word atom comes from a Greek word meaning something that cannot be split up any further. Today, we know this isn’t true. In theory, if you had a knife small and sharp enough, you could chop an atom of gold into bits and you’d find smaller things inside. But then you’d no longer have the gold: you’d just have the bits. All atoms are made from the same bits, which are called subatomic particles («sub» means smaller than and these are particles smaller than atoms). So if you chopped up an atom of iron, and put the bits into a pile, and then chopped up an atom of gold, and put those bits into a second pile, you’d have two piles of very similar bits—but there’d be no iron or gold left.
What are the parts of an atom?
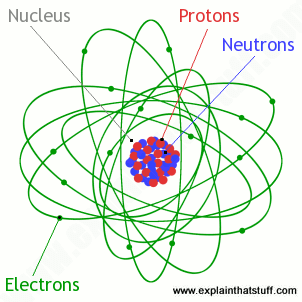
Most atoms have three different subatomic particles inside them: protons , neutrons , and electrons . The protons and neutrons are packed together into the center of the atom (which is called the nucleus ) and the electrons, which are very much smaller, whizz around the outside. When people draw pictures of atoms, they show the electrons like satellites spinning round the Earth in orbits. In fact, electrons move so quickly that we never know exactly where they are from one moment to the next. Imagine them as super-fast racing cars moving so incredibly quickly that they turn into blurry clouds—they almost seem to be everywhere at once. That’s why you’ll see some books drawing electrons inside fuzzy areas called orbitals .
Artwork: Atoms contain protons and neutrons packed into the central area called the nucleus, while electrons occupy the space around it. In simple descriptions of the atom, we often talk about electrons «orbiting» the nucleus like planets going around the Sun or satellites whizzing around Earth, although that’s a huge oversimplification. Note also that this picture isn’t drawn to scale! Most of an atom is empty space. If an atom were about as big as a baseball stadium, the nucleus would be the size of a pea in the very center and the electrons would be somewhere on the outside edge.
What makes an atom of gold different from an atom of iron is the number of protons, neutrons, and electrons inside it. Cut apart a single atom of iron and you will find 26 protons and 30 neutrons clumped together in the nucleus and 26 electrons whizzing around the outside. An atom of gold is bigger and heavier. Split it open and you’ll find 79 protons and 118 neutrons in the nucleus and 79 electrons spinning round the edge. The protons, neutrons, and electrons in the atoms of iron and gold are identical—there are just different numbers of them. In theory, you could turn iron into gold by taking iron atoms and adding 53 protons, 88 neutrons, and 53 electrons to each one. But if that were as easy as it sounds, you can bet all the world’s chemists would be very rich indeed!
But let’s suppose you could turn atoms into other atoms very simply. How would you make the first few chemical elements? You’d start with the simplest atom of all, hydrogen (symbol H), which has one proton and one electron, but no neutrons. If you add another proton, another electron, and two neutrons, you get an atom of helium (symbol He). Add a further proton, another electron, and two more neutrons, and you’ll have an atom of the metal lithium (symbol Li). Add one proton, one neutron, and one electron and you get an atom of beryllium (symbol Be).
See how it works? In all atoms, the number of protons and the number of electrons is always the same. So nitrogen has 7 protons and 7 electrons, calcium has 20 protons and 20 electrons, and tin has 50 protons and 50 electrons. The number of neutrons is very roughly the same as the number of protons, but sometimes it’s rather more. So bromine has 35 protons and 35 electrons, but 45 neutrons. Platinum has 78 neutrons, 78 electrons, and 117 neutrons. The number of protons in an atom is called the atomic number and it tells you what type of atom you have. An atomic number of 1 means the atom is hydrogen, atomic number 2 means helium, 3 means lithium, 4 is beryllium, and so on. The total number of protons and neutrons added together is called the relative atomic mass . Hydrogen has a relative atomic mass of 1, while helium’s relative atomic mass is 4 (because there are two protons and two neutrons inside). In other words, an atom of helium is four times heavier than an atom of hydrogen, while an atom of beryllium is nine times heavier.
What is the Periodic Table?
Suppose you make a list of the chemical elements in order of their atomic number (how many protons they have), starting with hydrogen (H). You’ll find that elements with similar chemical properties (how they react with things) and physical properties (whether they’re metals or non-metals, how they conduct heat and electricity, and so on) occur at regular intervals— periodically , in other words. If you rearrange your list into a table so similar atoms fall underneath one another, you get a diagram like this, which is called the Periodic Table . The columns are called groups and the rows are called periods.
Artwork: The Periodic Table of the elements.
So what? Atoms in a certain group (column) tend to have similar properties. So, for example, the red column on the right contains the Noble Gases (helium, neon, argon, krypton, and so on), which are relatively unreactive. The pink column on the left contains the alkali metals (lithium, sodium, potassium, and so on), which are relatively reactive metals (you probably know that some of them react violently with water, for example, to produce explosive hydrogen gas). If you know where a certain element sits in the table, and you know a little bit about the properties of the elements above, below, and either side, you can often figure out what the properties of that element will be.
How do atoms make molecules and compounds?
Atoms are a bit like people: they usually prefer company to being alone. A lot of atoms prefer to join up with other atoms because they’re more stable that way. So hydrogen atoms don’t exist by themselves: instead, they pair up to make what is called a molecule of hydrogen. A molecule is the smallest amount of a compound : a substance made from two or more atoms.
Some people find molecules and compounds confusing. Here’s how to remember the difference. If you join two different chemical elements together, you can often make a completely new substance. Glue two atoms of hydrogen to an atom of oxygen and you’ll make a single molecule of water. Water is a compound (because it’s two different chemical elements joined together), but it’s also a molecule because it’s made by combining atoms. The way to remember it is like this: compounds are elements joined together and molecules are atoms joined together.
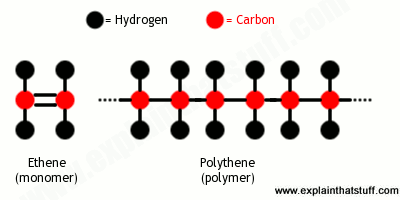
Not all molecules are as small and simple as water. Molecules of plastics, for example, can be made of hundreds or even thousands of individual atoms joined together in incredibly long chains called polymers . Polythene (also called polyethene or polyethylene) is a very simple example of this. It’s a polymer made by repeating a basic unit called a monomer over and over again—just like a coal train made by coupling together any number of identical trucks, one after another:
What are isotopes?
To complicate things a bit more, we sometimes find atoms of a chemical element that are a bit different to what we expect. Take carbon, for example. The ordinary carbon we find in the world around us is sometimes called carbon-12. It has six protons, six electrons, and six neutrons, so its atomic number is 6 and its relative atomic mass is 12. But there’s also another form of carbon called carbon-14, with six protons, six electrons, and eight neutrons. It still has an atomic number of six, but its relative atomic mass is 14. Carbon-14 is more unstable than carbon-12, so it’s radioactive : it naturally disintegrates, giving off subatomic particles in the process, to turn itself into nitrogen. Carbon-12 and carbon-14 are called isotopes of carbon. An isotope is simply an atom with a different number of neutrons that we’d normally expect to find.
How do atoms make ions?
Atoms aren’t just packets of matter: they contain electrical energy too. Each proton in the nucleus of an atom has a tiny positive charge (electricity that stays in one place). We say it has a charge of +1 to make everything simple (in reality, a proton’s charge is a long and complex number: +0.00000000000000000016021892 C, to be exact!). Neutrons have no charge at all. That means the nucleus of an atom is effectively a big clump of positive charge. An electron is tiny compared to a proton, but it has exactly the same amount of charge. In fact, electrons have an opposite charge to protons (a charge of −1 or −0.00000000000000000016021892 C, to be absolutely exact). So protons and electrons are a bit like the two different ends of a battery: they have equal and opposite electric charges. Since an atom contains equal number of protons and electrons, it has no overall charge: the positive charges on all the protons are exactly balanced by the negative charges on all the electrons. But sometimes an atom can gain or lose an electron to become what’s called an ion . If it gains an electron, it has slightly too much negative charge and we call it a negative ion; it it loses an electron, it becomes a positive ion.
Artwork: A lithium atom (Li) forms a positive ion (Li + ) by «losing» an electron. A fluorine atom (F) forms a negative ion (F − ) by gaining an electron.
What’s so good about ions? They’re very important in many chemical reactions. For example, ordinary table salt (which has the chemical name sodium chloride) is made when ions of sodium join together with ions made from chlorine (which are called chloride ions). A sodium ion is made when a sodium atom loses an electron and becomes positively charged. A chloride ion forms in the opposite way when a chlorine atom gains an electron to become negatively charged. Just like two opposite magnet poles, positive and negative charges attract one another. So each positively charged sodium ion snaps onto a negatively charged chloride ion to form a single molecule of sodium chloride. When compounds form through two or more ions joining together, we call it ionic bonding . Most metals form their compounds in this way.
The electrical charge that ions have can be useful in all sorts of ways. Ions (as well as electrons) help to carry the electricity through batteries when you connect them into a circuit.
How many atoms are there in something?
If atoms are so tiny, there must be zillions and zillions of them in all the things around us. but how many are there, exactly ?
Chemists have a handy way of talking amount these vast numbers of atoms—by using the rather unusual word mole. A mole of something (anything) has exactly 6.022 × 10 23 particles in it, which is a short way of saying 602,200,000,000,000,000,000,000 or 602 billion trillion. This strange amount is called Avogadro’s number (or Avogadro’s constant) after Italian chemist Amedeo Avogadro (1776–1856), who thought up the idea. Avogadro’s original hypothesis was that a certain volume of any gas will contain the same number of molecules as the same volume of any other gas providing both gases are at the same temperature and pressure.
So how much is a mole? When we’re talking about atoms, a mole is the relative atomic mass in grams. So a mole of carbon is 12g, because carbon’s relative atomic mass is 12, and it contains 620 billion trillion atoms. A mole of aluminum is 27g, because aluminum’s relative atomic mass is 27. A mole of aluminum also contains 620 billion trillion atoms.
We can also use moles to talk about molecules. A mole of a compound contains 602 billion trillion molecules. A molecule of water has a relative molecular mass of 18 (that’s 16 for the oxygen atom, plus two hydrogens, making 18). A mole of water weighs 18g and contains 620 billion trillion molecules.
Photo: A mole of any substance contains the same number of elementary particles (atoms, molecules, ions, electrons, or anything else). Here you can see 18g of water, 12g of carbon, 63g of copper, and 27g of aluminum. Each of these is a mole and contains 602 billion trillion atoms (or molecules, in the case of water). Photo courtesy of National Institute of Standards and Technology Digital Collections, Gaithersburg, MD 20899.
How do we know atoms exist?
Artwork: Molecules are built from atoms: In the early 19th century, English chemist John Dalton (1766–1844) realized that atoms join together in simple ratios. Water forms when two hydrogens snap onto one oxygen. Chemical reactions like this make sense if the elements exist as simple building blocks: atoms, in other words.
If we can’t see atoms, how do we know they’re there? That’s a very good question! Science is all about evidence, so what evidence do we have that atoms really exist? It comes in a variety of different forms:
- Chemists have long known that when we combine different elements in chemical reactions, the ingredients join in simple ratios. So, for example, in water we know that there are twice as many hydrogen atoms as oxygen atoms (H 2 O), making a ratio of 2:1. In salt (sodium chloride) there are equal numbers of sodium and chlorine atoms (NaCl), so the ratio is 1:1. We can easily explain this if chemical elements really exist as simple particles (atoms, in other words), which snap together like building blocks.
- Some substances are radioactive: they naturally split into simpler substances and give off tiny particles or energy in the process. Again, this makes sense if atoms exist and they’re built from smaller particles (protons, neutrons, and electrons).
- Scientists can split big atoms into smaller ones. In one very famous series of experiments in the early 20th century, a team led by Ernest Rutherford (a New-Zealand-born physicist) fired particles at atoms and watched what happened. This showed how the bits were arranged inside a typical atom (with the nucleus at the center).
- We have plenty of evidence for tiny particles called electrons: they power things like electricity and magnetism. An English physicist named J.J. Thomson discovered electrons in 1897. This discovery helped scientists to realize that atoms are made of even smaller bits.
- Unlike these earlier scientists, we can actually see atoms; just look at the photo of sulfur atoms up above! Seeing that picture would have delighted Rutherford, Thomson, and the other pioneers of atomic science. Now, scientists are even starting to see inside atoms. Thanks to the development of really powerful electron microscopes, we can peer deep inside things at their internal atomic structure. In 2013, for example, scientists used a quantum microscope to produce the first picture of the inside of a hydrogen atom. Amazing!
There’s plenty more evidence where that came from, but this will do for starters. It shows us that our theory of what atoms are and how they are built is a very good one: the theory agrees with the things we see around us in the world and it’s confirmed by many different kinds of evidence. It’s not a complete theory, however: we still have an enormous amount to learn about atoms and the bits and pieces lurking inside them!
A brief history of atoms
Who discovered the atom, how, and when? Let’s quickly nip back through history.
- 450 BCE: Ancient Greek philosophers Leucippus and Democritus became the first people to propose that matter is made of atoms.
- 1661: Anglo-Irish chemist Robert Boyle (1627–1691) suggested that chemical elements were the simplest forms of matter.
- 1789: Frenchman Antoine Lavoisier (1743–1794), widely known as the «father of modern chemistry,» set out a list of chemical elements (which he defined as substances that can’t be broken down through a chemical reaction). This was an important stepping stone on the way to the full Periodic Table.
- 1803: English scientist John Dalton (1766–1844) published the atomic theory of matter. He realized each chemical element was made up of atoms.
- 1815: English physician William Prout (1785–1850) suggested the weights of different elements are simple multiples of the weight of a hydrogen atom—not quite true, but another important clue to understanding how atoms are made.
- 1869: Building on the insights of Lavoisier, Dalton, Prout and others, a Russian chemist called Dmitri Mendeleyev (1834–1907) found a logical way of organizing the chemical elements with a neat structure called the Periodic Table.
- 1896: French physicist Henri Becquerel (1852–1908) accidentally discovered radioactivity.
- 1917: New Zealand-born English physicist Ernest Rutherford (1871–1937) «split» the atom: he proved that atoms are made of smaller particles, eventually concluding they had a heavy, positively charged nucleus and a largely empty area around them.
- 1919: British physicist Francis Aston (1852–1908) discovered a large number of atomic isotopes using mass spectrometry.
- 1938: German physicists Otto Hahn (1879–1978) and Fritz Strassmann (1902–1980) achieved the first nuclear fission (splitting up of heavy atoms to make lighter ones).
- 1945: The United States dropped atomic bombs on the Japanese cities of Hiroshima and Nagasaki.
- 1960s–1970s: Particle physicists figured out how several fundamental forces hold small, «subatomic» particles together to make atoms. Their ideas gradually became known as the Standard Model.
- 2013: Scientists used a quantum microscope to take the first pictures inside a hydrogen atom.
Find out more
On this website
On other sites
- The Particle Adventure: One of the best simple websites explaining atoms and the world inside them.
- Structure of Matter: This very good interactive slideshow from the Nobel Prize website explains, in 22 slides, all about atoms and the other particles inside them. [Archived via the Wayback Machine.]
- Dark matter and dark energy: Most of the «stuff» in the universe isn’t conventional matter or energy, as we’ve always conceived it: it’s actually «dark matter» and «dark energy.» This NASA website explains what these things are and how they relate to conventional matter and energy.
Articles
- Ultrasensitive Microscope Reveals How Charging Changes Molecular Structures by Dexter Johnson, IEEE Spectrum, July 11, 2019. What happens to the structure of molecules when they gain or lose electric charges?
- Scientists Took an M.R.I. Scan of an Atom by Knvul Sheikh, The New York Times, July 1, 2019. Scientists from IBM Almaden and the Institute for Basic Sciences in Seoul discover what happens when you look at atoms in a body scanner.
- Do Not Adjust Focus. Those Blobs Are Atoms by Kenneth Chang, The New York Times, October 5, 2004. The quest to take pictures of an atom continues!
Books for younger readers
- Can you feel the force? by Richard Hammond. Dorling Kindersley, 2015. Includes a short introduction to atoms, quantum theory, and particle physics.
- The Periodic Table by Adrian Dingle. Oxford University Press, 2014.
- Atoms and Molecules by Chris Woodford and Martin Clowes. Rosen, 2012. (Previously published by Blackbirch.) One of my own books, this charts the history of atomic theory from ancient times to modern.
- Splitting the Atom by Alan Morton. Evans, 2005.A brief history of atom theory.
- How to split the atom by Hazel Richardson. Oxford University Press, 1999. A zany, fun guide that will appeal to lovers of the Horrible Science series.
- Eyewitness: Matter by Christopher Cooper. Dorling Kindersley, 1992. A drier, but solid introduction; good for school projects.
Books for older readers
- Atom by Piers Bizony. Icon, 2017. The story of how scientists came to understand atoms.
- The Elements: A Visual Exploration of Every Known Atom in the Universe by Theodore Gray. Black Dog & Leventhal, 2012. An entertaining trip through the Periodic Table.
- Mr. Tompkins in Paperback by George Gamov. Cambridge, 2012. A very vivid introduction to the world inside atoms from one of the 20th-century’s most creative physicists. Suitable for older teens and above.
- The Fly in the Cathedral by Brian Cathcart. Farrar, Straus and Giroux, 2005. Excellent, easy-to-understand account of how Ernest Rutherford and his team figured out the structure of atoms. Also published in paperback by Penguin.
- Six Easy Pieces by Richard Feynman. Penguin, 1998. This book is by no means as «easy» as its title suggests, but the final chapter does contain a pithy explanation of quantum theory and its various puzzles that people with a basic grounding in physics can hope to understand.
Videos
- What is a Higgs boson?: Don Lincoln, a scientist at Fermilab, explains the hottest question in subatomic science—in terms most of us can understand!
- What is antimatter?: Another good simple explanation from Don Lincoln.
- How J.J. Thomson discovered the electron: This is a great little video that explains how scientists such as Thomson came to the conclusion that electrons must be charged particles inside atoms.
Please do NOT copy our articles onto blogs and other websites
Articles from this website are registered at the US Copyright Office. Copying or otherwise using registered works without permission, removing this or other copyright notices, and/or infringing related rights could make you liable to severe civil or criminal penalties.
Text copyright © Chris Woodford 2007, 2021. All rights reserved. Full copyright notice and terms of use.
Follow us
Rate this page
Please rate or give feedback on this page and I will make a donation to WaterAid.
Save or share this page
Press CTRL + D to bookmark this page for later or tell your friends about it with:
Источник